[最新] heterogeneous catalyst and homogeneous catalyst 280165-What is homogeneous catalysis
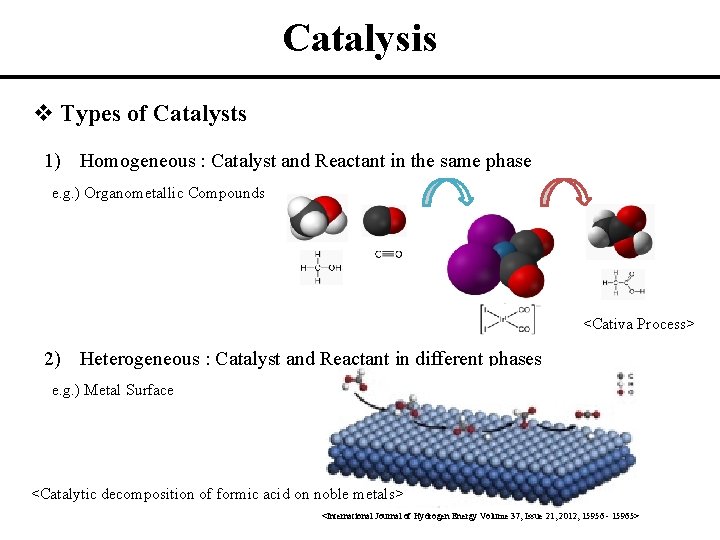
Homogeneous and heterogeneous catalysis Activity is the ability of the catalyst to accelerate a chemical reaction The degree can be as high as 100 times in certain reactions A catalytic cycle processes in which the reactant and catalyst undergo several transformations before making theHeterogeneous metal catalysts rather than homogeneous ones are recommended for industrial applications after considering their performance in activity, separation, and recycling 1Homogeneous and Heterogeneous Catalysis The phenomenon of catalysis can be divided into two main types – homogeneous and heterogeneous catalysis, on the bases of the number of phases present in the reaction mixture (A phase is a homogeneous part of a system)
Www Ethz Ch Content Dam Ethz Special Interest Chab Icb Van Bokhoven Group Dam Coursework Catalysis 17 Homogeneous Heterogeoenous Catalysis Mesoporousmaterials Pdf
What is homogeneous catalysis
What is homogeneous catalysis-In heterogeneous catalysis, catalysts provide a surface to which reactants bind in a process of adsorption In homogeneous catalysis, catalysts are in the same phase as the reactants Enzymes are biological catalysts that produce large increases in reaction rates and tend to be specific for certain reactants and products Catalysts are conventionally divided into two categories homogeneous and heterogeneous Enzymes , natural biological catalysts, are often included in the former group, but because they share some properties of both but exhibit some very special properties of their own, we will treat them here as a third category



Http Www Ijetsr Com Images Short Pdf 1496 1500 Ieteb410 Ijetsr Pdf
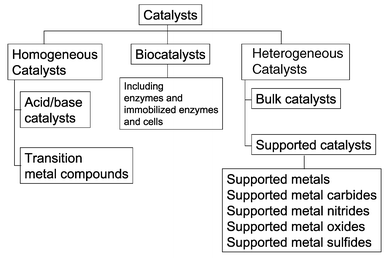
Homogeneous catalyst Accordingly a primary object of this invention is to provide a technique of converting normally homogene ous transition metal complex catalysts to heterogeneous phase catalysts of substantially equal catalytic activity as the normally homogeneous phase "parent" catalyst Another object of this invention is to provide aOur homogeneous catalysts can be used in a wide range of chemical transformations Catalysts in varying purities and concentrations Due to their high selectivity, precious metal chemicals are often the first choice as heterogeneous catalysts for a wide variety of reactions, including hydrogenation, hydrosilation, isomerization and carbonylationCatalysts and their associated catalytic reactions come in three main types homogeneous catalysts, heterogeneous catalysts and biocatalysts (usually called enzymes)
Homogeneous and heterogeneous catalysis With the use of catalysis in chemistry, we can realise reactions under milder conditions and obtain a maximum yield of the desired product Wageningen Food & Biobased Research develops new, catalytic processes for the conversion of biomass into highquality chemicals The development of new, sustainable Surface organometallic chemistry is an area of heterogeneous catalysis which has recently emerged as a result of a comparative analysis of homogeneous and heterogeneous catalysis The chemical industry has often favored heterogeneous catalysis, but the development of better catalysts has been hindered by the presence of numerous kinds of active Catalyst poisoning tests using mercury and dibenzoa,ecyclooctene show that dynamic Pd nanoclusters maintain their catalytic activity, which demonstrate their combined features of homogeneous and
Heterogeneous versions of homogeneous catalysts can often be produced and may have certain advantages in use particularly on a commercial scale The catalytic chemistry of supported rhodium compounds for the carbonylation of methanol is described and the behaviour of heterogeneous and homogeneous operation compared in order to illustrate the relations between the two catalystCatalysts can be divided into two main types heterogeneous and homogeneous In a heterogeneous reaction, the catalyst is in a different phase from the reactants In a homogeneous reaction, the catalyst is in the same phase as the reactantsHeterogeneous Heat Transfer might be an issue due to the different heat capacities of reactants and catalystCatalyst Separation Homogeneous The separation of the products from the catalyst is generally expensive, the only exception being in biphasic catalysis Heterogeneous The separation of the products from the catalyst is usually




Navigating Through The Maze Of Homogeneous Catalyst Design With Machine Learning Trends In Chemistry




Heterogeneous Vs Homogeneous Catalysts Advantages
Many catalytic processes are known in which the catalyst and the reactants are not present in the same phase—that is, state of matter These are known as heterogeneous catalytic reactions They include reactions between gases or liquids or both at the surface of a solid catalyst Since the surface is the place at which the reaction occurs, it generally is prepared in ways that produce large surface areas per unit of catalystBoth hydrogenation with homogeneous and heterogeneous catalysts is covered Typically, additives (alcohols, amines or aminoalcohols) are used to transform CO 2 into intermediates, which can further be reduced into methanolHomogeneous and Heterogeneous Catalysis We model catalysis, spectroscopy and photochemistry of molecular systems containing transition metals and also catalytic phenomena involving metal or metaloxide clusters attached to a support, like, for example, a




Comparison Of Heterogeneous And Homogeneous Catalysis Download Table




C 4 1 Compare The Modes Of Action Of Homogeneous And Heterogeneous Catalysts Youtube
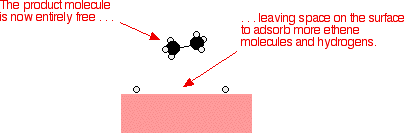
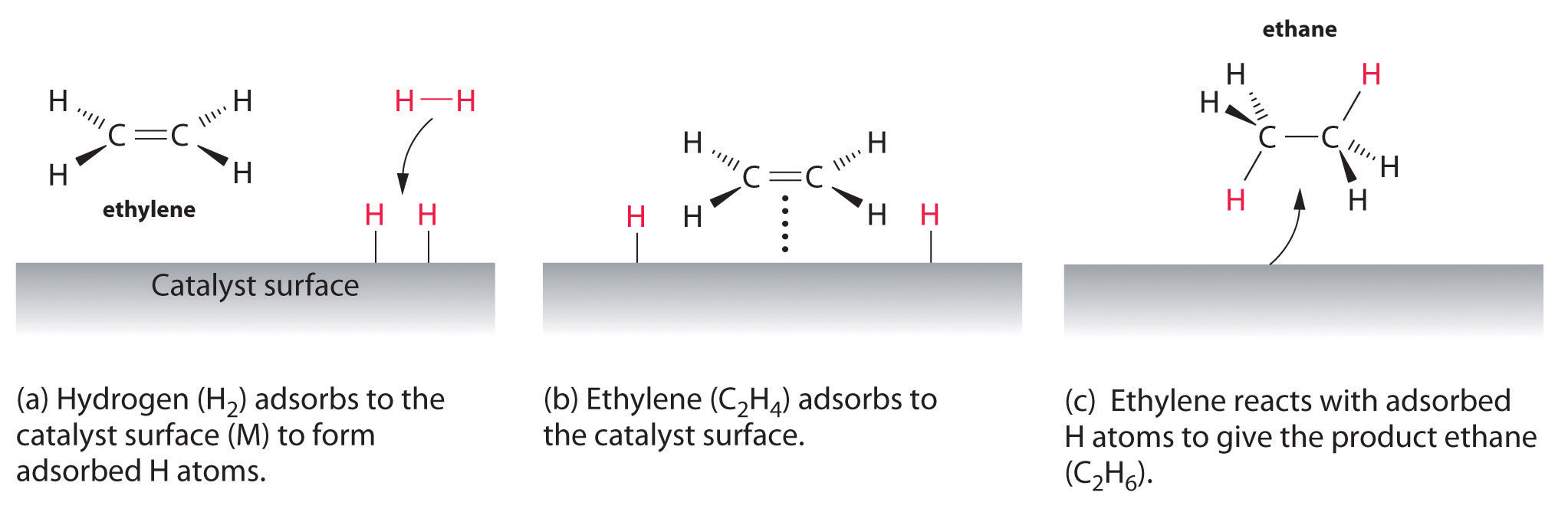
This video provides a basic introduction into homogeneous and heterogeneous catalysts A Homogeneous catalyst exists in the same phase as the reactants andHeterogeneous catalysts are just a tethered version of a homogeneous catalyst At the microscopic, bondbreaking, bondmaking level, usually only a few atoms close to the bond being broken/formed are critical Big practical difference is that it is easy to separate/immobilize heterogeneous catalyst Also, heterogeneous catalysts have some diffusionHeterogeneous catalysts are chemical catalysts whose physical phase is different from the physical phase of the reactants and/or products that take part in the catalyzed chemical reaction Typically, solid phase heterogeneous catalysts are employed in order to facilitate the chemical reaction between two gaseous reactants




Difference Between Homogeneous And Heterogeneous Catalyst Compare The Difference Between Similar Terms
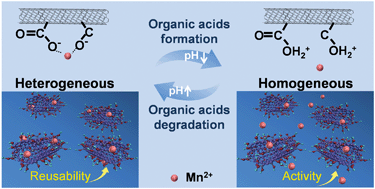



Performing Homogeneous Catalytic Ozonation Using Heterogeneous Mn2 Bonded Oxidized Carbon Nanotubes By Self Driven Ph Variation Induced Reversible Desorption And Adsorption Of Mn2 Environmental Science Nano Rsc Publishing
Polymer science has profited enormously from CuAAC by its simplicity, ease, scope, applicability and efficiency Basic principles of the CuAAC are reviewed with a focus on homogeneous and heterogeneous catalysts, ligands, anchimeric assistance, and basic chemical principles Recent developments of ligand design and acceleration are discussedCatalysis is a multidisciplinary activity which is reflected in this book The editors have chosen a combination of basic disciplines homogeneous catalysis by metal complexes is treated jointly with heterogeneous catalysis with metallic and nonmetallic solids The main theme of the book is the molecular approach to industrial catalysisHomogeneous catalysis refers to reactions where the catalyst is in the same phase as the reactants, principally in solution In contrast, heterogeneous catalysis describes processes where the catalysts and substrate are in distinct phases, typically solidgas, respectively




Comparison Of Homogeneous And Heterogeneous Catalysts Download Scientific Diagram
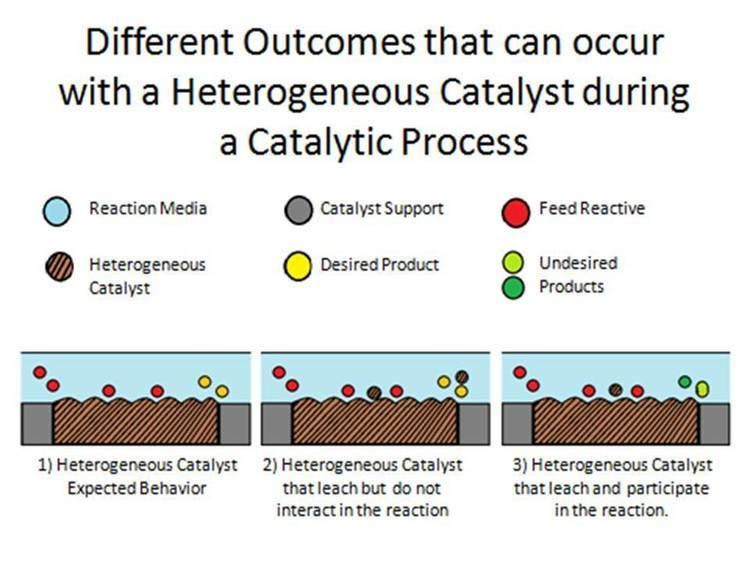



Heterogeneous Catalysis Alchetron The Free Social Encyclopedia
A homogeneous catalyst is a catalyst that is in the same phase as the reactants, while a heterogeneous catalyst is in a different phase than theCatalysts Homogeneous & Heterogeneous in a Snap! One example of a heterogeneous catalyst is the catalytic converter in gasoline or dieselfueled cars Likewise, what are the 3 types of catalysis?
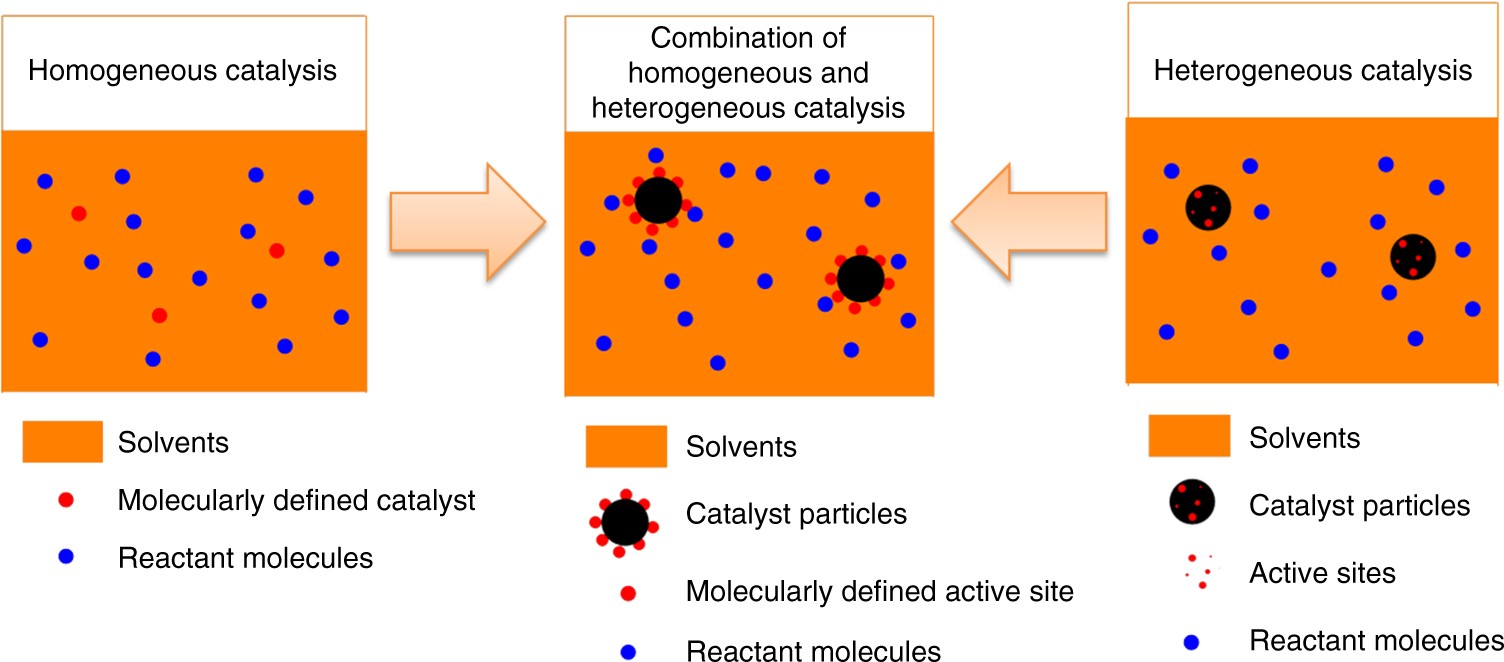



Synthesis Of A Molecularly Defined Single Active Site Heterogeneous Catalyst For Selective Oxidation Of N Heterocycles Nature Communications



Q Tbn And9gctrvbbisy7ikjyh35 8msk3jkudkzizuszlwrim5fn3f2i54ygk Usqp Cau
SO using 3 NO as a homogeneous catalyst, which occurs in the gaseous phase On the other hand, most of the processes using homogeneous catalysts occur in a liquid phase whereas for the heterogeneous catalysts, the catalyst is usually in a solid form, and the reaction occurs either in the liquid or gaseous phase The fact that the catalysts is in aThe catalyst is not behaving like a conventional homogeneous molecular catalyst but more like the metallic active sites exploited in heterogeneous catalysts 'We still get singlesite In this respect, heterogeneous singlemetalsite catalysts combine features of homogeneous and heterogeneous catalysis Ideally, in metalbased catalysts, any metal atom constitutes a single low




Difference Between Homogeneous Catalysis And Heterogeneous Catalysis Surface Chemistry Youtube




Crossing The Divide Between Homogeneous And Heterogeneous Catalysis In Water Oxidation Pnas
There are two types of catalysisHomogeneous Catalysis In this type of catalysis, both catalyst and reactant are present in the same phase ExampleIn the oxidation of SO 2 to SO 3, Nitric oxide (NO) is used as a catalyst Here, catalyst (NO) and reactants (SO 2 and O 2) are present in the same phase (ie gaseous phase)Unlock the full Alevel Chemistry course at http//bitly/2ZDDPTi created by Ella Buluwela, Chemistry experIntroducing amino groups as substituents to molecular catalysts is a promising approach towards improving capture and reduction of CO 2 This review explores recently developed stateoftheart molecular catalysts with a focus on heterogeneous and homogeneous amine molecular catalysts for electroreduction of CO 2




Catalysis Boundless Chemistry




Reshaping The Cathodic Catalyst Layer For Anion Exchange Membrane Fuel Cells From Heterogeneous Catalysis To Homogeneous Catalysis Ren 21 Angewandte Chemie Wiley Online Library
Abstract Interest in catalysis by metal nanoparticles (NPs) is increasing dramatically, as reflected by the large number of publications in the last five years This field, "semiheterogeneous catalysis", is at the frontier between homogeneous and heterogeneous catalysis, and progress has been made in the efficiency and selectivity of reactions and recovery Homogeneous catalysts have many attractive properties, such as high selectivities However, many homogeneous catalytic systems cannot be commercialized because of difficulties associated with separating the products from the catalyst Recent approaches to tackling this problem are reviewed and comparedIn chemistry, heterogeneous catalysis is catalysis where the phase of catalysts differs from that of the reactants or productsThe process contrasts with homogeneous catalysis where the reactants, products and catalyst exist in the same phase Phase distinguishes between not only solid, liquid, and gas components, but also immiscible mixtures (eg oil and water), or anywhere
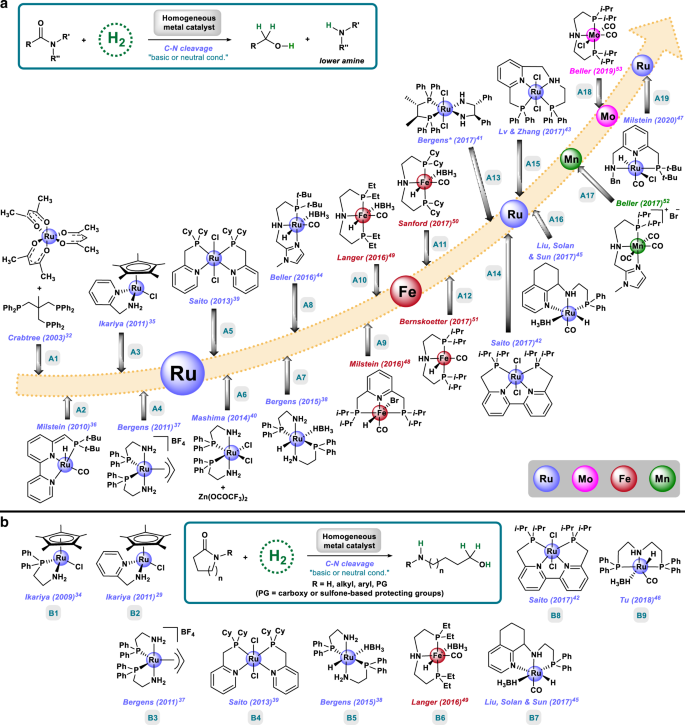



Homogeneous And Heterogeneous Catalytic Reduction Of Amides And Related Compounds Using Molecular Hydrogen Nature Communications




Catalysis Boundless Chemistry
Homogeneous catalysis takes place when the catalyst and the other reactants are all dissolved in the same solution Heterogeneous catalysis typically involves the use of a catalyst that is insoluble, or perhaps only weakly soluble, in the solution in which the reaction takes place Thus, in heterogeneous catalysis, the catalyst and solution may form a suspension, or the catalyst mayHomogeneous gold catalysts have high activity, enantioselectivity, and wellcharacterized structures, but heterogeneous gold systems show easier separation of the catalyst from the products, possible catalyst recycling, and adaptation to continuous flow processes @article{osti_, title = {Single Atom Catalysis An Analogy between Heterogeneous and Homogeneous Catalysts}, author = {Yuk, Simuck F and Collinge, Gregory B and Nguyen, Manh Thuong and Lee, Mal Soon and Glezakou, VassilikiAlexandra and Rousseau, Roger J}, abstractNote = {In recent years, enormous efforts have been invested to improve the atomefficiency of metal catalysts




Explain The Difference Between A Homogeneous And Heterogeneous Catalyst Give An Example Of Each Youtube




Homogeneous Catalyst Chander Jaswal Assistant Professor Chemistry Homogeneous
Homogeneous catalysts are those which exist in the same phase (gas or liquid) as the reactants, while heterogeneous catalysts are not in the same phase as the reactants Typically, heterogeneous catalysis involves the use of solid catalysts placed in a liquid reaction mixture Catalysis Note the lowered activation energy of the catalyzed pathwayTel 31 46 b Department of Chemistry, Joseph Black Building, University of Glasgow, Glasgow, UK G12 8QQHomogeneous and heterogeneous catalysis in industry Johannes G de Vries a and S David Jackson b a DSM Innovative Synthesis BV, PO Box 18, 6160 MD Geleen, The Netherlands Email HansJGVriesde@dsmcom;



Types Of Catalysis




Ppt Starter 1 Definition Of Catalysts 2 Difference Between Homogeneous And Heterogeneous Catalyst Powerpoint Presentation Id
A homogeneous catalyst is a catalyst that is capable of dissolving in solution, because it by definition is in the same phase as the rest of the reactants in the solution Here are the principles of homogeneous catalysts that I see in my textbook (Inorganic Chemistry, Shriver, Atkins, Ch 25) PROS Homogeneous catalysts are effective at being highly selective towardsA catalyst is a substance that changes (usually increases) the rate of a reaction without undergoing any permanent chemical alteration Catalysts are generally divided into two types, those that are in the same phase as the reactants (homogeneous catalysts) and those that belong to a different phase (heterogeneous catalysts)Catalysts can be divided into two types, homogeneous or heterogeneous, depending on the reaction phase that they occupy Homogeneous catalysts are those that occupy the same phase as the reaction mixture (typically liquid or gas), while heterogeneous catalysts occupy a different phase Generally, heterogeneous catalysts are solid compounds that are added to liquid or gas




Organometallic Catalysis




1 Give Three Reasons Why Heterogeneous Catalysts Are Chegg Com
Heterogeneous catalysts based on supported gold nanoparticles have become among the most efficient and selective catalysts for a series of organic reactions, including aerobic oxidations, hydrogenation of nitro groups, Fenton degradation, among others This Chapter provides a historical perspective of how gold catalysis has developed in parallel to the progress of Nanoscience and Nanotechnology, highlighting the importance of the particle size distribution in gold catalysisA homogeneous catalyst is referred to as a catalytic reaction where the catalyst has the same phase with the reactant In short, it is a soluble catalyst in a solution Homogeneous catalyst generally provides short reaction time and needs a few amounts of catalyst in the catalytic system owing to it can contact with reactant adequately compareThe sulfonated carbonbased solid acid catalyst was thus designed to be an active, stable and reusable solid acid as an environmentally benign replacement for homogeneous catalyst
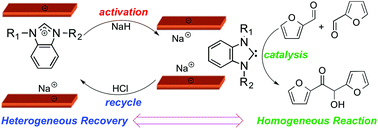



An Interchangeable Homogeneous Heterogeneous Catalyst System For Furfural Upgrading Green Chemistry Rsc Publishing



Www Osti Gov Servlets Purl
Representation of a tethered complex on a supported metal (TCSM) The metal complex, M(L′) 3 X, is a homogeneous complex catalyst and is supported on a heterogeneous metal catalyst, which consists of metal particles on a silica support The metals of the complex and of the heterogeneous catalyst may be combinations of two platinum group metalsHomogeneous vs heterogeneous catalysis Dr habil Marko Hapke 3 3 Heterogeneous Catalysis Homogeneous Catalysis Catalyst and reactant(s) are in the same phase Catalyst and reactant(s) are in different phases Definitions General features Different reaction phases possible „classic"




Relationship Among Sacs Homogeneous Catalysts And Heterogeneous Catalysts Download Scientific Diagram
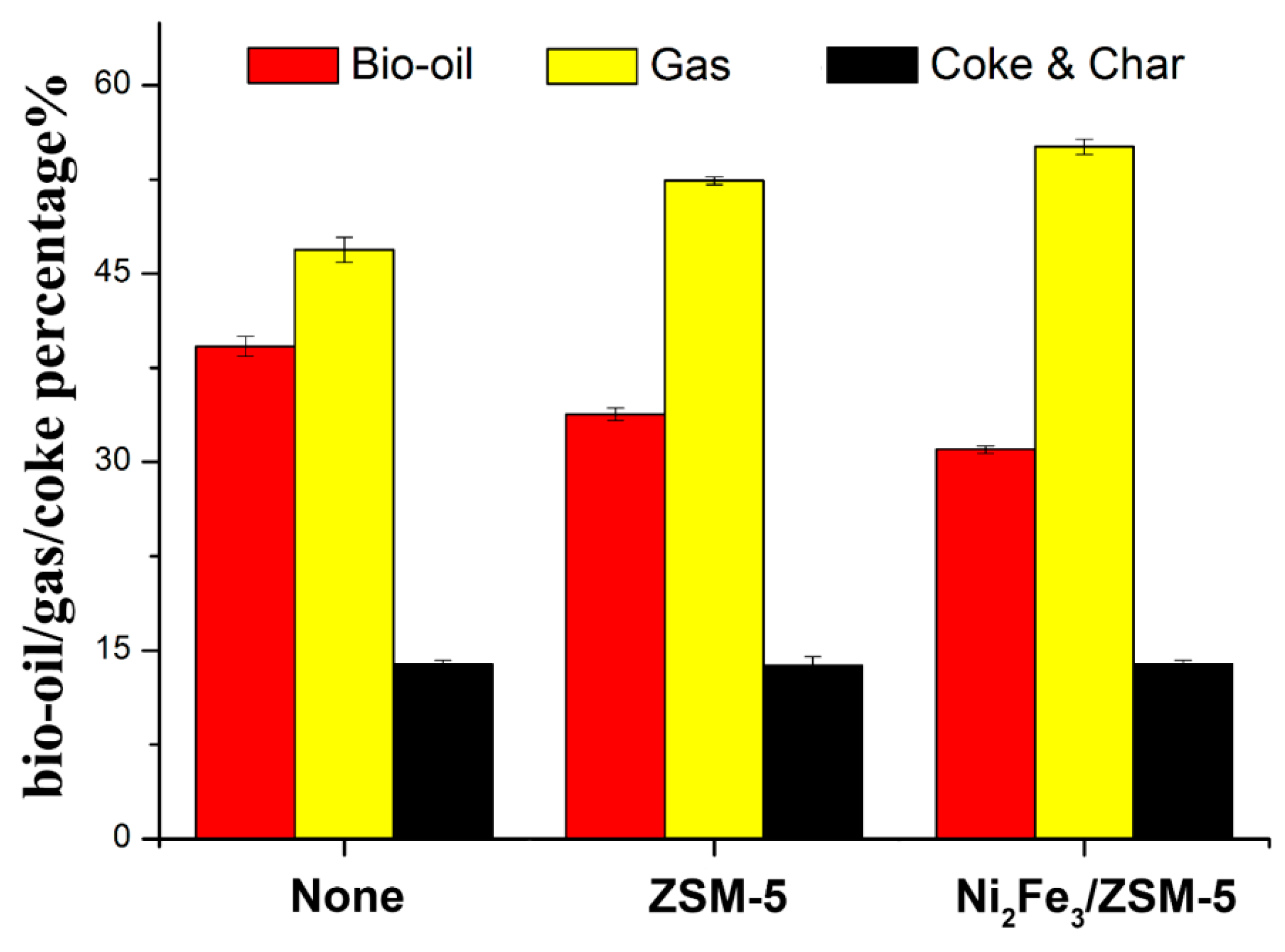



Catalysts Free Full Text Homogeneous And Heterogeneous Catalysis Impact On Pyrolyzed Cellulose To Produce Bio Oil Html




Review Of Catalytic Transesterification Methods For Biodiesel Production Intechopen
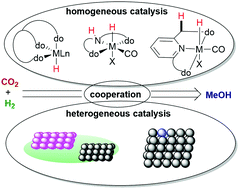



Homogeneous And Heterogeneous Catalysts For Hydrogenation Of Co2 To Methanol Under Mild Conditions Chemical Society Reviews Rsc Publishing




A Review Of The Problem Of Distinguishing True Homogeneous Catalysis From Soluble Or Other Metal Particle Heterogeneous Catalysis Under Reducing Conditions Sciencedirect



Bridging Homogeneous And Heterogeneous Catalysis By Heterogeneous Single Metal Site Catalysts Nature Catalysis
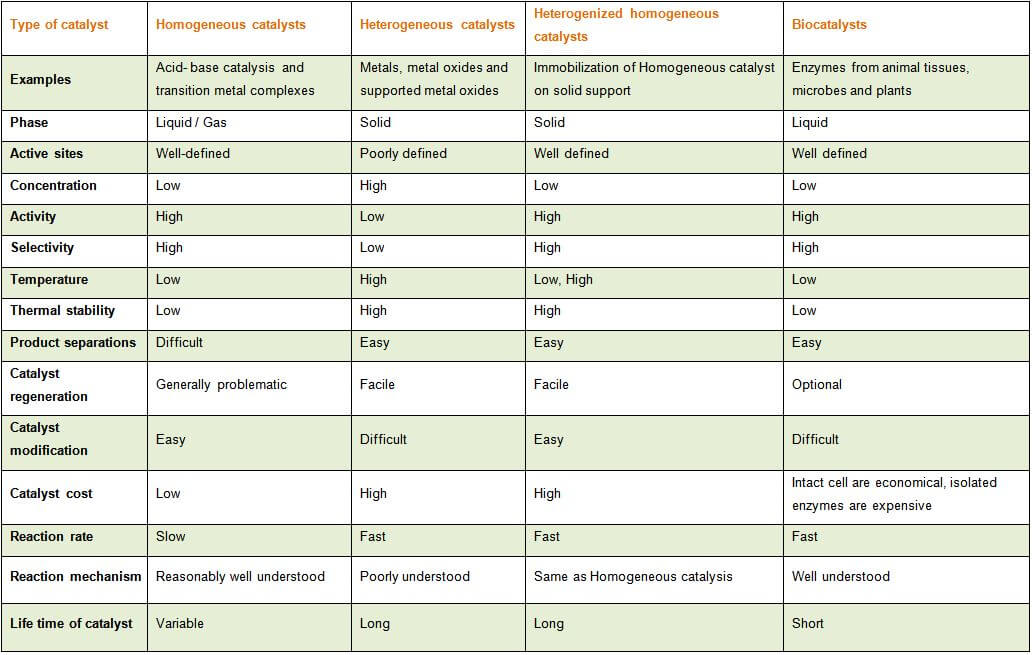



An Overview Of Different Types Of Catalysts Legal Advantage



Presenting My Work At The Netherlands Catalysis And Chemistry Conference March 19 European Training Network For The Sustainable Zero Waste Valorisation Of Critical Metal Containing Industrial Process Residues
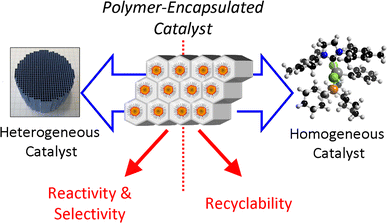



Polymer Encapsulated Metallic Nanoparticles As A Bridge Between Homogeneous And Heterogeneous Catalysis Springerlink



Www Ethz Ch Content Dam Ethz Special Interest Chab Icb Van Bokhoven Group Dam Coursework Catalysis 17 Homogeneous Heterogeoenous Catalysis Mesoporousmaterials Pdf




Introduction To Catalysis Chemistry Ppt Video Online Download




1 Homogeneous Vs Heterogeneous Catalysts Download Table




Heterogeneous Homogeneous Catalysts Video Lesson Transcript Study Com




Kinetics Theory Of Catalytic Mechanisms Heterogeneous Catalysis Homogeneous Catalyzed Reaction Examples Advanced A Level Gce Revision Notes



Single Atom Catalyst Based On Homogeneous Catalysis Prototype For Co2 Transformation Developed 催化与新材料研究室
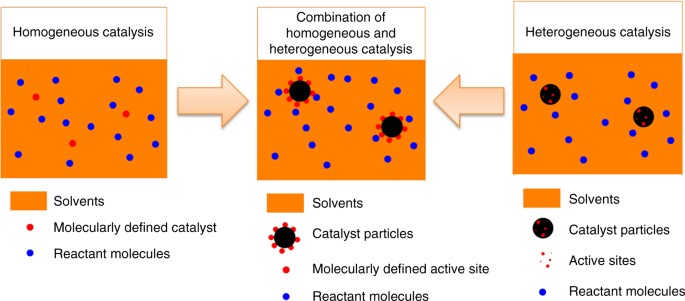



Synthesis Of A Molecularly Defined Single Active Site Heterogeneous Catalyst For Selective Oxidation Of N Heterocycles Nature Communications



2




Asymmetric Heterogeneous Catalysis




Homogeneous Borotungstic Acid And Heterogeneous Micellar Borotungstic Acid Catalysts For Biodiesel Production By Esterification Of Free Fatty Acid Sciencedirect




Neet Ug Homogeneous And Heterogeneous Catalyst In Hindi Offered By Unacademy



Q Tbn And9gctrvbbisy7ikjyh35 8msk3jkudkzizuszlwrim5fn3f2i54ygk Usqp Cau



Types Of Catalysis Homogeneous Catalysis Heterogeneous Catalysis Positive Catalysis Negative Catalysis Induced Catalysis Acid Base Catalysis
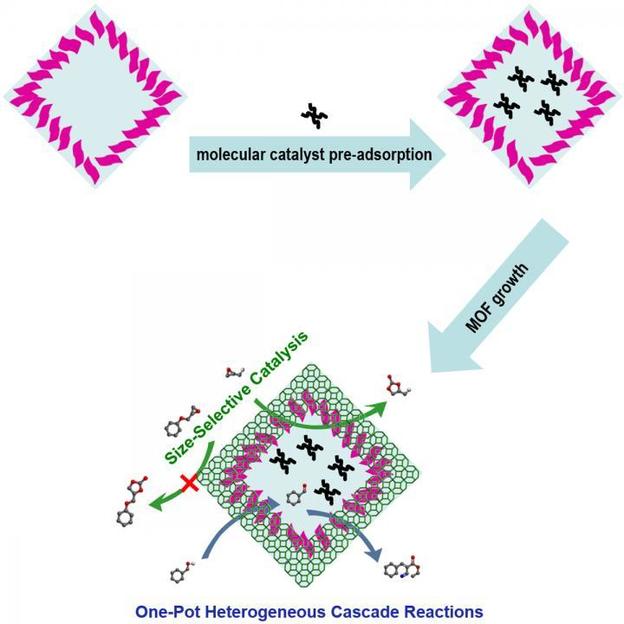



Lab In Hollow Mof Capsules Beyond Integration Of Homogeneous And Heterogeneous Catalysis
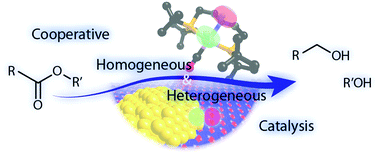



Heterogeneous And Homogeneous Catalysis For The Hydrogenation Of Carboxylic Acid Derivatives History Advances And Future Directions Chemical Society Reviews Rsc Publishing




Homogeneous Catalysis Wikipedia



Homogeneous Versus Heterogeneous Catalysis Download Scientific Diagram
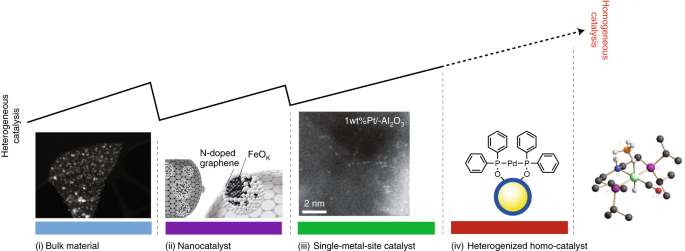



Bridging Homogeneous And Heterogeneous Catalysis By Heterogeneous Single Metal Site Catalysts Nature Catalysis




Chapter 13 8 Catalysis Chemistry Libretexts



Sciendo Com Pdf 10 1515 Eces 18 0001
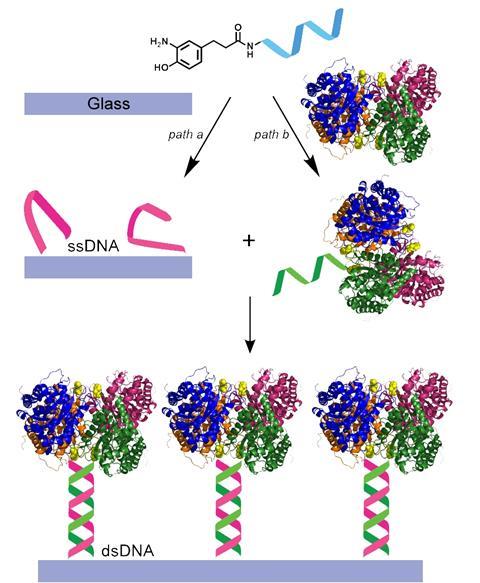



Combining Homogeneous And Heterogeneous Catalysis Feature Chemistry World



1
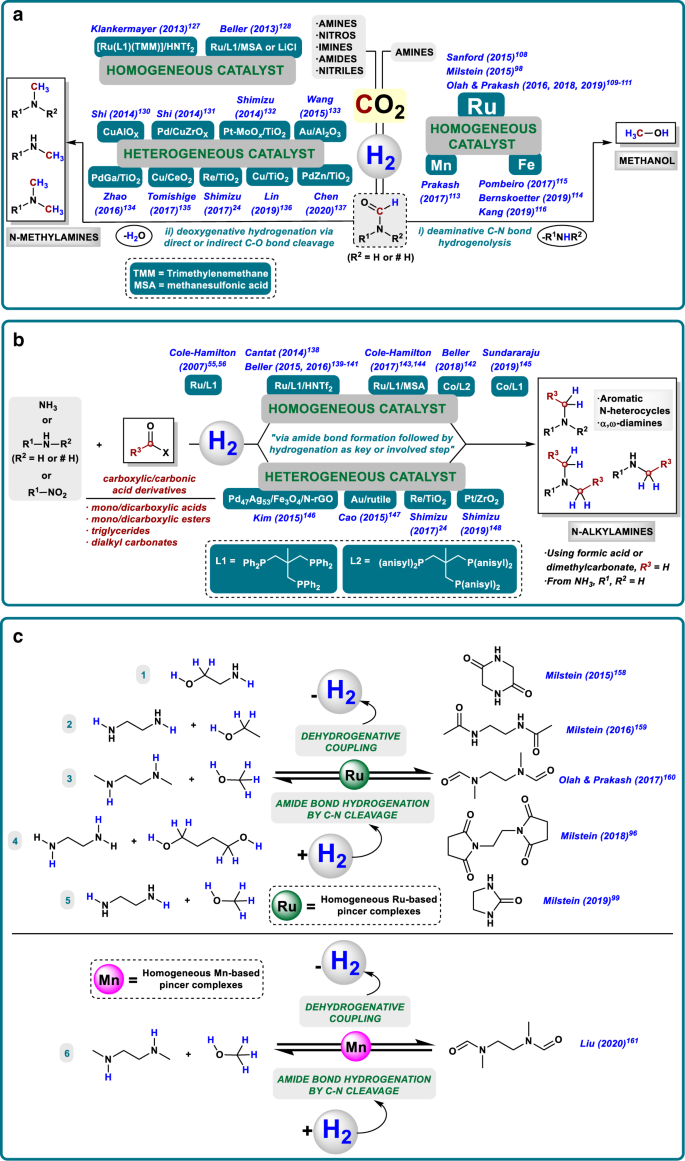



Homogeneous And Heterogeneous Catalytic Reduction Of Amides And Related Compounds Using Molecular Hydrogen Nature Communications




Homogeneous Catalyst An Overview Sciencedirect Topics




Kinetics Theory Of Catalytic Mechanisms Heterogeneous Catalysis Homogeneous Catalyzed Reaction Examples Advanced A Level Gce Revision Notes
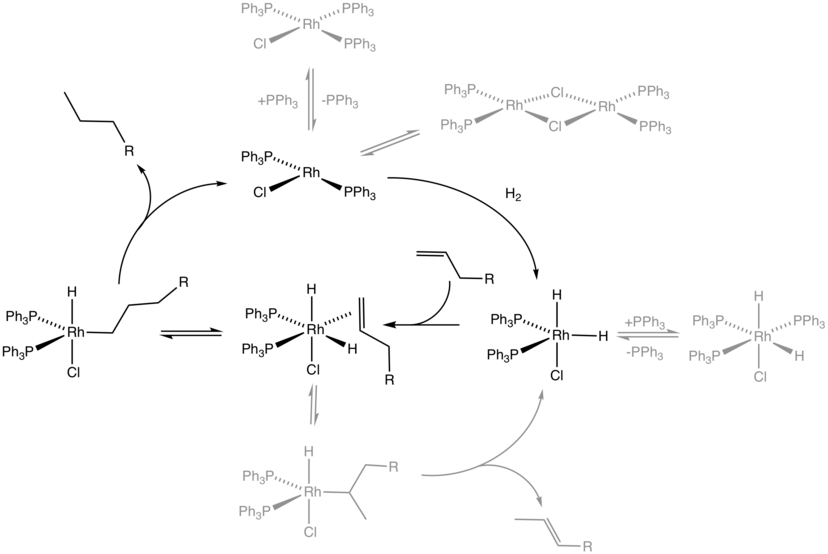



Homogeneous Catalysis Wikiwand




Homogeneous And Heterogeneous Catalysis W3spoint




Comparison Of Biodiesel Production Between Homogeneous And Heterogeneous Base Catalysts Scientific Net




Homogeneous And Heterogeneous Catalysis Impact On Pyrolyzed Cellulose To Produce Bio Oil Researcher An App For Academics
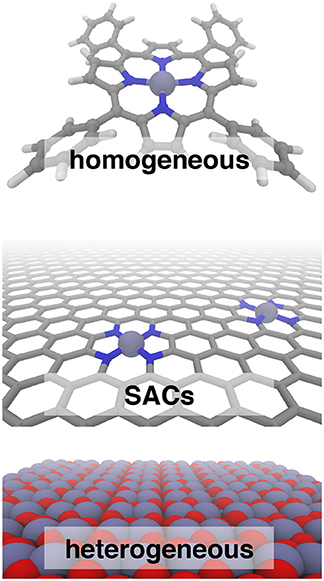



Frontiers Bridging The Homogeneous Heterogeneous Divide Modeling Spin For Reactivity In Single Atom Catalysis Chemistry




Heterogeneous Catalysis Wikipedia




1 Schematic Diagram Of The Reaction Pathes In Homogeneous Download Scientific Diagram



Catalytic Mechanisms Of Hydrogen Evolution With Homogeneous And Heterogeneous Catalysts Energy Environmental Science Rsc Publishing




Homogeneous Heterogeneous Catalysts Cie A Level Chemistry 19 21 Notes




Homogeneous Catalysis Wikipedia The Free Encyclopedia Catalysis Chemical Kinetics




Differences Between Homogeneous And Heterogeneous Ef Processes Download Table




Homogeneous Heterogeneous Catalysts 19 21 Cie As Chemistry Notes



Http Www Ijetsr Com Images Short Pdf 1496 1500 Ieteb410 Ijetsr Pdf




Advantages And Disadvantages Of Homogeneous And Heterogeneous Catalysts Download Scientific Diagram




Homogeneous Catalysis Introduction Ppt Video Online Download




Research Brochure Catalysis And Magnetic Resonance Group By Cebcambridge Issuu



Hal Sorbonne Universite Fr Hal Document




Industrial Applications Of Homogeneous Catalysis A Mortreux Springer
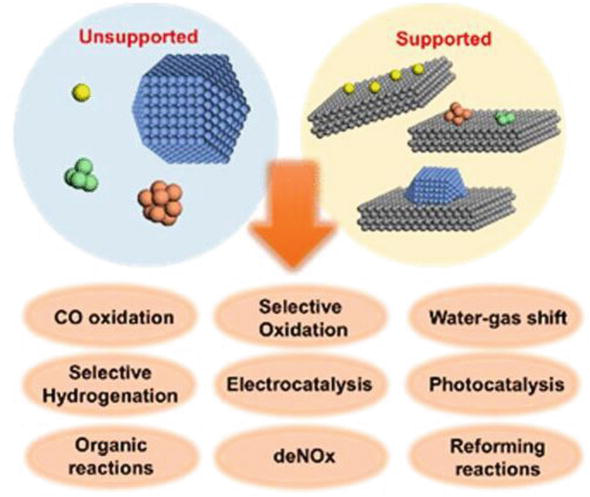



Heterogeneous Catalytic Process For Wastewater Treatment Intechopen



Heterogeneous Catalysis And Catalyst Recycling All About Drugs



Heterogeneous Catalysis Solid State Physics 141 A Dohyung



Heterogeneous Catalysis And Catalyst Recycling All About Drugs



Homogeneous Catalysis For The Conversion Of Biomass And Biomass Derived Platform Chemicals Catalysis Science Technology Rsc Publishing



Snapshots Of A Working Catalyst Possibilities And Limitations Of In Situ Spectroscopy In The Field Of Heterogeneous Catalysis Chemical Communications Rsc Publishing



Types Of Catalysis




Chemical Kinetics Made Easy Homogeneous And Heterogeneous Catalysts Episode 13 Youtube



Catalysis



Heterogeneous Catalyst Goes Enzymatic



Homogeneous Catalysis Qs Study



How Can Heterogeneous Catalysts Differ From Homogeneous Catalysts Quora




Homogeneous Vs Heterogeneous Catalysts Basic Introduction Youtube
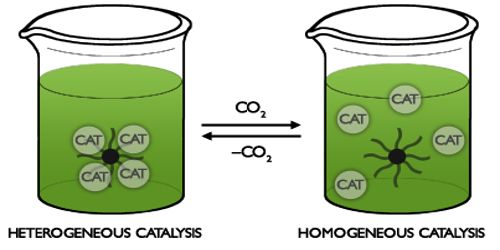



Homogeneous Catalysis Qs Study




1 Homogeneous Vs Heterogeneous Catalysts Download Table




Design And Use Of Nanostructured Single Site Heterogeneous Catalysts For The Selective Transformation Of Fine Chemicals Topic Of Research Paper In Chemical Sciences Download Scholarly Article Pdf And Read For Free On



23 5 Features Of Homogeneous Catalysis A Catalyst




Heterogeneous Catalysis Wikipedia




Comparative Investigation Of Homogeneous And Heterogeneous Bronsted Base Catalysts For The Isomerization Of Glucose To Fructose In Aqueous Media Sciencedirect
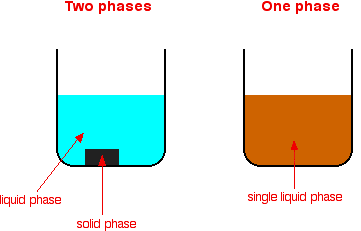



Types Of Catalysis




Homogeneous Catalysis Catalysis Heterogeneous Catalysis




Ch3010 A What Are The Advantages And Disadvantages Chegg Com




Pdf Encyclopedia Of Life Support Systems Eolss Homogeneous And Heterogeneous Catalysis Semantic Scholar



1




Combining Homogeneous And Heterogeneous Catalysis Feature Chemistry World



Immobilization Of Jacobsen Type Catalysts On Modified Silica




Bridging Heterogeneous And Homogeneous Catalysis Concepts Strategies And Applications Wiley




Single Atom Catalysis Bridging The Homo And Heterogeneous Catalysis Sciencedirect
コメント
コメントを投稿